Products and services
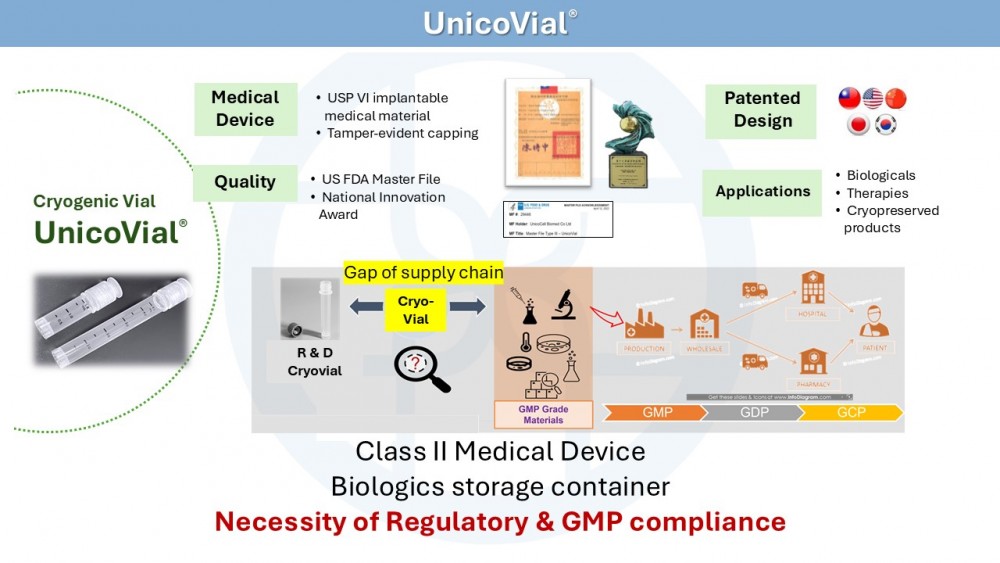
Cryogenic Vial (Class 2 Medical Device)

UnicoVial®, a patented cryogenic vial, obtained approval of TFDA Class 2 medical device, received Acknowledgment letter of DMF (Drug Master File) from US FDA and won the 19th National Innovation Award.
UnicoVial® is ideal for storage of cell products and biological samples under ultra-low temperature, with a stopper invention specifically designed for clinical use.
Product Description








UnicoVial® Learn more
UnicoVial®, the only cryogenic vial certified as Class 2 medical device by Taiwan FDA.
UnicoVial® is suitable for the storage of biologics, microbial samples, and biological samples at ultra-low temperatures.
UnicoCell strives for excellence and delivers a high quality cryogenic container UnicoVial® for clinical use. For further information, please contact info@unicocell.com


